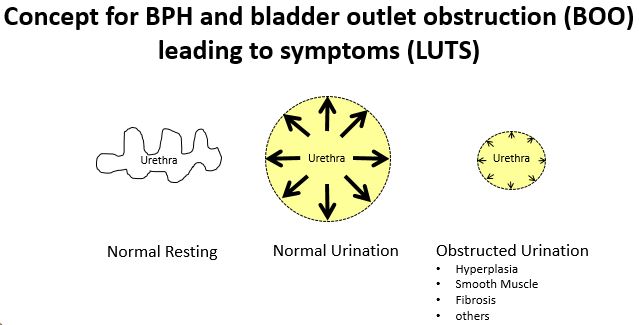
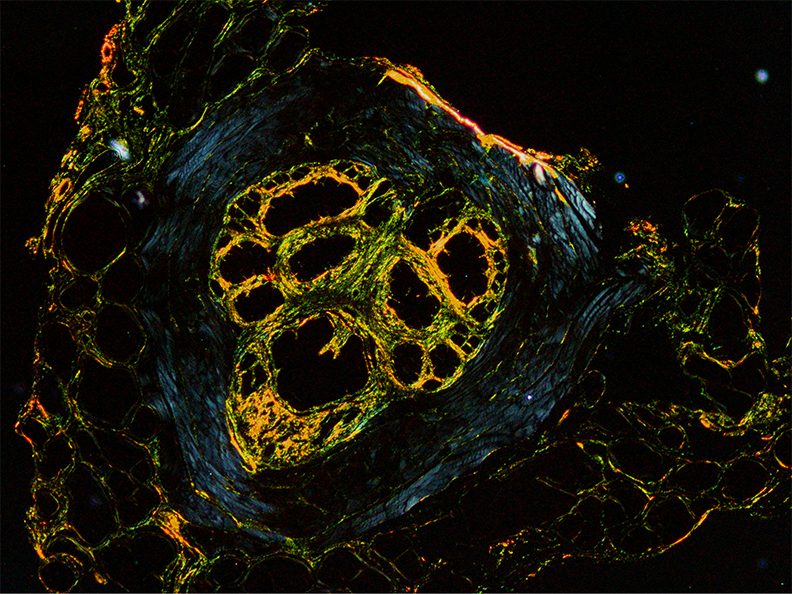
Project 1. Estrogens stimulate prostatic collagen synthesis to drive fibrosis and LUTD.
Estrogens drive prostate fibrosis, hence voiding dysfunction, through expression of collagen in prostate stromal cells. This project identifies collagen as a mediator of prostatic fibrosis and lower urinary tract dysfunction. We will test whether collagen deposition alone is sufficient to induce voiding dysfunction in mice, independent of prostate urethral hyperplasia. We will also determine if therapeutic anti-fibrotic therapies are effective in the treatment of prostate fibrosis and associated voiding dysfunction. Broader ongoing themes of our research is to determine if patient stratification for prostate fibrosis/LUTS is possible. Collaboratively, we endeavor to identify urinary proteins and metabolites as predictive biomarkers for appropriate medical or surgical therapy. Furthermore, we will develop novel imaging methods for prostate fibrosis and disease in models of voiding dysfunction. Our research strives to decrease BPH/LUTS associated morbidity and increase lifespan globally.
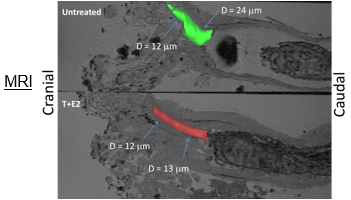
Project Leaders:
William Ricke, PhD
Credentials: Professor of Urology; O’Brien Center Director; UWMF-Professor of Urologic Research; Director of Research
Email:
rickew
Phone: (608) 265-3202
Diego Hernando, PHD
Credentials: Assistant Professor, Radiology and Medical Physics; O’Brien Center Project 1 Co-Investigator, UW-Madison
Email:
dhernando
Phone: (608) 265-7590
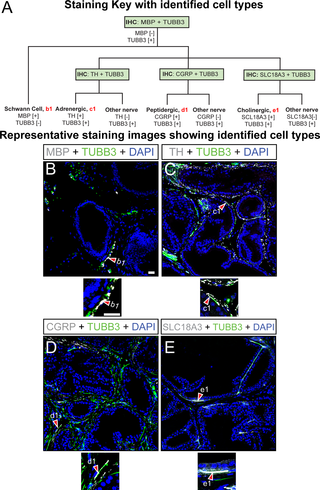
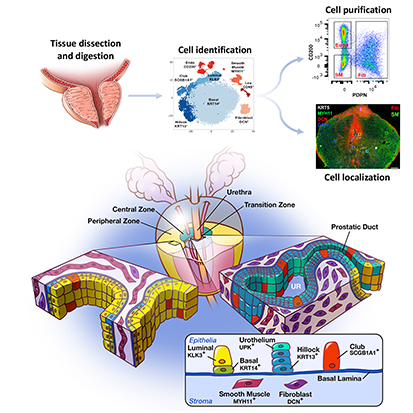
Project 2. CTGF drives voiding dysfunction through expression of collagen in periurethral SRD5A2+ fibroblasts.
This project identifies a cell type and a factor responsible for pathological collagen production by: (1) conducting single cell RNA Sequencing on human prostate tissues from 20 to 80 year old men, (2) performing genetic cell lineage analysis using a novel mouse strain, (3) testing connective tissue growth factor as a prostatic fibrosis-inducing protein in mice, and (4) testing whether an investigational new drug that targets connective tissue growth factor is effective at reducing prostatic collagen content and voiding dysfunction in a mouse with prostatic inflammation. The overarching hypothesis is that inflammation causes steroid five alpha reductase type 2 (SRD5A2)-positive prostatic fibroblasts to proliferate and synthesize connective tissue growth factor and collagen, thereby increasing prostatic and peri-urethral stiffness, bladder outlet resistance, and voiding dysfunction. The project contributes a foundational resource by expanding the human prostatic single cell RNA sequencing database we created.
Project Leaders:
Chad Vezina, PhD
Credentials: O’Brien Center Associate Director; Associate Professor of Comparative Biosciences
Email:
chad.vezina
Phone: (608) 890-3235
Doug Strand, PHD
Credentials: Assistant Professor of Urology; O’Brien Center Project 2 Co-leader, UT-Southwestern
Email:
Douglas.Strand
Phone: (214) 648-6096
Project 3. Persistence of an IL-4/IL-13 autocrine loop promotes fibrosis-mediated urinary voiding dysfunction.
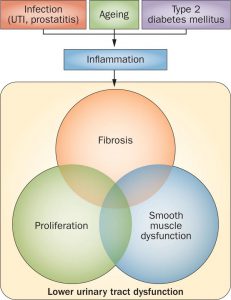
This project hypothesizes that some peri-urethral stromal cell populations establish an IL-4/IL-13 axis that self-perpetuates, induces myofibroblast phenoconversion and survival, and upregulates collagen accumulation, thereby promoting consequent urinary voiding dysfunction. To test this hypothesis, Project 3 will: 1) determine whether signaling through the IL-4 receptor creates a STAT6- and GATA-3-mediated positive feedback loop, 2) elucidate the mechanisms through which the IL-4/IL-13 axis promotes myofibroblast phenoconversion and concordant collagen expression, 3) determine whether IL-4 pathologically represses myofibroblast apoptosis and thereby promotes myofibroblast survival, and 4) test whether the IL-4/IL-13 signaling axis promotes lower urinary tract fibrosis and urinary voiding dysfunction in vivo. The results of these preclinical studies will elucidate previously unknown interleukin-mediated molecular and cellular mechanisms that promote lower urinary tract fibrosis and dysfunction. Using JAK/STAT inhibitors to therapeutically target this potentially self-perpetuating mechanism may ‘break the cycle’ and successfully treat recalcitrant peri-urethral prostatic fibrosis contributing to voiding dysfunction development and progression.
Project Leaders:
Jill Macoska, PhD
Credentials: Alton J. Brann Distinguished Professor in Science and Mathematics; Professor of Biological Sciences; Director, Center for Personalized Cancer Therapy
Email:
Jill.Macoska
Phone: (617) 287-5783
Paul Marker, PhD
Credentials: Professor of Pharmaceutical Sciences; Associate Dean of Research; Vice Chair of Pharmaceutical Sciences
Email:
paul.marker
Phone: Phone (608) 890-2150
The Biomedical Research Core

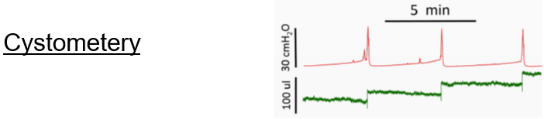
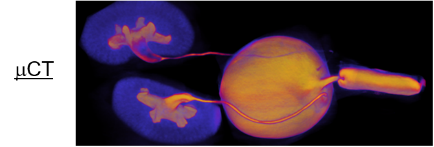
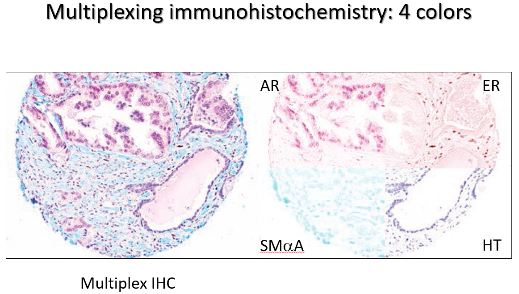 The Biomedical Research Core is the first NIH-funded Core for rodent urinary function testing (RUFT) and its services are available to all researchers on a collaborative or fee for service basis. All three projects leverage Core B resources. The Core is responsible for some of our most impactful community resources, including publicly distributed software for objective, unbiased and efficient analysis of void spot assay which was heralded as a feature story in AJP Renal. The Core also created and publicly distributed software for analyzing uroflowmetry data. We are refining ultrasonography as a means to assess lower urinary tract structure and urine passage through the prostatic urethra and are using MRI in conjunction with contrast agents to assess urinary tract structure. We want the entire community to benefit from this Core, and therefore in the proposed funding period will develop a short course open to all investigators within and outside our Center, which provides training in mouse urinary function testing and data analysis.
The Biomedical Research Core is the first NIH-funded Core for rodent urinary function testing (RUFT) and its services are available to all researchers on a collaborative or fee for service basis. All three projects leverage Core B resources. The Core is responsible for some of our most impactful community resources, including publicly distributed software for objective, unbiased and efficient analysis of void spot assay which was heralded as a feature story in AJP Renal. The Core also created and publicly distributed software for analyzing uroflowmetry data. We are refining ultrasonography as a means to assess lower urinary tract structure and urine passage through the prostatic urethra and are using MRI in conjunction with contrast agents to assess urinary tract structure. We want the entire community to benefit from this Core, and therefore in the proposed funding period will develop a short course open to all investigators within and outside our Center, which provides training in mouse urinary function testing and data analysis.
Project Leaders:
Dale Bjorling, DVM
Credentials: O’Brien Center Biomed Core Co-Director; Professor of Surgical Sciences; Associate Dean of Research School of Veterinary Medicine
Email:
dale.bjorling
Phone: (608) 263-4808
Kimberly Keil Stietz, PhD
Credentials: O’Brien Center Biomed Core Co-Director; Assistant Professor, Comparative Biosciences
Email:
kkeil
Phone: (608) 265-2879
Administration Core (Core A)
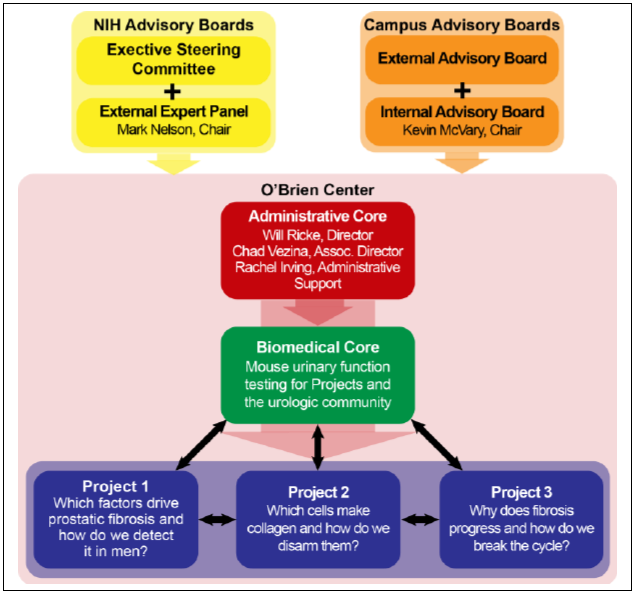 The Admin Core leads and promotes interactions for the Center in the following manner:
The Admin Core leads and promotes interactions for the Center in the following manner:
- Facilitates interactions between research projects and the biomedical core
- Promotes collaboration of Center members other U54s and P20 centers nationwide
- Provides means for outstanding training venues
- K12 at UW and nationwide
- graduate and postdoctoral training programs at UW, UTSW, UMass (e.g. T32)
- summer programs in undergraduate urological research
- undergraduate research
- Enables novel research interactions through pilot projects from new and established investigators
Project Leaders:
William Ricke, PhD
Credentials: Professor of Urology; O’Brien Center Director; UWMF-Professor of Urologic Research; Director of Research
Email:
rickew
Phone: (608) 265-3202
Chad Vezina, PhD
Credentials: O’Brien Center Associate Director; Associate Professor of Comparative Biosciences
Email:
chad.vezina
Phone: (608) 890-3235








